Requirements for placing new cosmetic ingredients on the market in China
The National Medical Products Administration (NMPA) is China’s central authority responsible for the supervision, registration, filing and safety oversight of cosmetics, cosmetic ingredients and related products. Under the Cosmetic Supervision and Administration Regulation (CSAR), effective from January 1, 2021, the NMPA ensures that all cosmetic products placed on the Chinese market comply with strict safety, quality, and regulatory requirements. Non‑compliance with NMPA requirements under CSAR can lead to financial penalties and it means the product has not legally cleared regulatory checks to enter the China market.
Beyond cosmetics oversight by the NMPA, products placed on the Chinese market must also meet wider chemical compliance obligations. This includes preparing China‑compliant Safety Data Sheets (SDS) and labels in line with national GHS requirements, as well as ensuring 24‑hour emergency response coverage within China, often provided through NRCC‑approved services. These obligations operate alongside CSAR requirements and support safe, compliant market entry.
Importance of NMPA
Through CSAR and its supporting regulations, the NMPA has modernized China’s cosmetic regulatory landscape by creating a more structured and transparent system for evaluating cosmetic products and ingredients. It has strengthened requirements for safety data, scientific assessment, and post market monitoring so that only ingredients and products meeting rigorous safety standards can enter and remain in the Chinese market. These reforms also offer clearer and more predictable compliance pathways for domestic and international companies, simplifying navigation through registration, notification, and ongoing safety obligations in China.
How to determine if a cosmetic ingredient requires notification or registration
Registration or notification of new cosmetic ingredients (including new uses) in China is submitted to the NMPA.
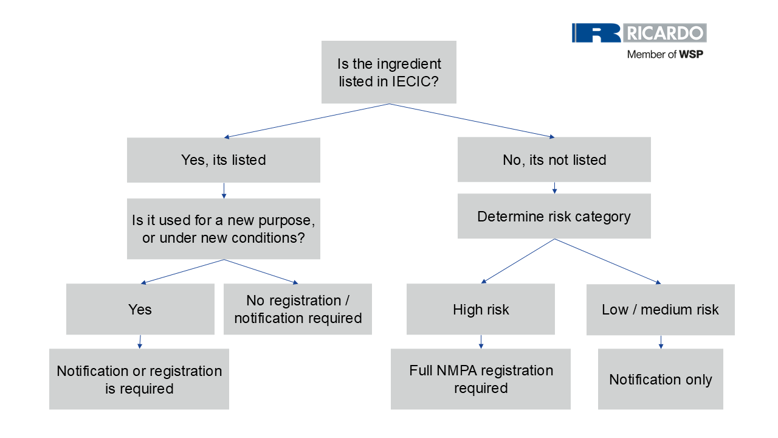
High-risk new cosmetic ingredients, such as preservatives, sunscreens, colorants, hair dyes, and whitening or spot correcting agents, are subject to full registration with the NMPA due to their functional impact and safety considerations.
Lower and medium risk new ingredients, on the other hand, only require notification, which involves a simplified filing process through the online system.
Regulatory approach for pure cosmetic ingredients vs cosmetic mixtures
Under China’s cosmetic regulatory framework, the requirements for new cosmetic ingredient (NCI) registration or notification remain the same regardless of whether the substance is supplied as a pure cosmetic ingredient or as part of a cosmetic mixture. What matters is solely whether any individual chemical substance within the material is considered new or existing according to CSAR definitions. A new cosmetic ingredient is defined as a natural or synthetic substance used for the first time in China, or an existing substance being used under new conditions such as a new purpose or changed dosage, as this triggers the need for registration or notification.
Therefore, mixtures themselves are not registered; only the individual components are assessed. If all ingredients in the mixture are already listed in the IECIC and used within permitted conditions, no premarket procedure is required. However, if a mixture contains even one component that qualifies as a new cosmetic ingredient, the company must submit the required dossier for that specific ingredient. This means the only practical difference when dealing with mixtures is that companies must check and report every component to confirm whether any of them meet the definition of a new cosmetic ingredient under CSAR.
Support for your business
If you are a manufacturer of chemicals or cosmetic ingredients and wish to place your products on the Chinese market, Ricardo can support you with:
- Full notification and registration of new cosmetic ingredients under China NMPA
- Preparation of China‑compliant Safety Data Sheets (SDS) and product labelling
- Provision and coordination of 24‑hour emergency response services within China (NRCC‑aligned)
- Support with chemical registrations and broader China regulatory compliance requirements
Don’t risk delays, rejected submissions or costly enforcement action in China; ensure your ingredients, documentation and emergency response measures meet every NMPA, CSAR, SDS and NRCC requirement.
Speak to our experts to secure fully compliant, confident and seamless access to the Chinese market
Global regulatory and compliance supportRicardo supports organisations with expert guidance on global chemical regulations, including REACH and REACH‑like regimes, SDS authoring and assurance, labelling, risk management, and compliance strategies, helping businesses navigate complex, evolving requirements to ensure product safety, legal market access and sustainable regulatory confidence. |
 |









